📞  — free, 24/7
— free, 24/7
 — free, 24/7
— free, 24/7
| Dosage | Package | Price per Dose | Price | |
|---|---|---|---|---|
| 0.1/0.3% | 5 bottles | C$17.60 | C$109.99 C$87.99 Best Price Popular | |
| 0.1/0.3% | 4 bottles | C$21.05 | C$105.20 C$84.16 | |
| 0.1/0.3% | 3 bottles | C$26.13 | C$98.03 C$78.42 | |
| 0.1/0.3% | 2 bottles | C$35.39 | C$88.46 C$70.77 | |
| 0.1/0.3% | bottle | C$51.64 | C$64.55 C$51.64 |
Disclaimer: This page provides general information about Tobradex for residents of Canada. It is not a substitute for medical advice from a doctor or pharmacist. Tobradex may require a prescription depending on local rules; always follow your healthcare professional’s instructions and the product leaflets.
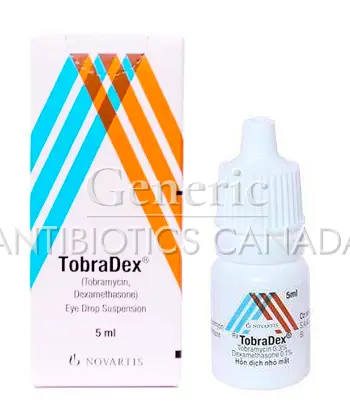
Tobradex is an eye medication that combines two active ingredients: tobramycin, an antibiotic, and dexamethasone, a corticosteroid. It is intended for use in the eye to treat certain infections and inflammation.
The medicine is supplied as an ophthalmic preparation for topical use on the eye. It is designed to be applied as eye drops or an eye suspension, under medical supervision.
It may be prescribed for adults and, in some cases, for children, depending on the doctor’s assessment. Tobradex is usually used for short periods under close supervision to control infection and swelling in the eye.
As with any medicine, Tobradex has safety considerations. Do not use Tobradex if you have a known allergy to tobramycin, dexamethasone, or any ingredient in the product. It should not be used to treat viral eye infections or fungal eye infections unless specifically advised by a clinician. Be cautious if you wear contact lenses; your doctor will advise whether you should remove lenses before use and when you may reinsert them.
In Canada, Tobradex may require a prescription depending on local rules. Follow the label directions and the guidance provided by your healthcare professional. If you have questions about whether Tobradex is right for you, speak with a pharmacist or physician.
Tobradex is used to treat eye infections caused by bacteria that are sensitive to tobramycin. It also helps reduce inflammation in the eye that accompanies certain infections or surgical procedures.
Common indications include conditions such as conjunctivitis and other superficial eye infections where inflammation is present. It may also be used after eye surgery to help control inflammation and promote healing, as advised by a clinician.
Because the product contains a steroid, Tobradex helps lessen swelling and irritation in the eye but should be used only under medical supervision. The combination is not appropriate for all eye conditions, and a clinician will assess whether this medication is suitable for your situation.
Never use Tobradex for conditions that are not clearly diagnosed or not approved for this medicine. If you are unsure whether your eye problem is due to an infection or inflammation that Tobradex can treat, consult a healthcare professional promptly. Always follow the product leaflet and your clinician’s instructions.
If Tobradex is not improving your eye symptoms or if symptoms worsen, contact your doctor for further evaluation. Do not extend the duration of use beyond what your clinician has recommended without advice.
The medicine combines two actions. Tobramycin fights bacteria by interfering with the production of bacterial proteins, helping to stop the infection from spreading. Dexamethasone reduces inflammation and swelling by dampening the body’s normal inflammatory responses.
Because Tobradex contains a corticosteroid, it can ease redness and irritation when used for a short period under supervision. The antibiotic component helps address bacterial causes of infection, while the steroid component helps with inflammation that may accompany the infection.
Because of the steroid part, Tobradex is not appropriate for all eye conditions, especially viral or fungal infections. It is important to use it only as directed by a healthcare professional and to monitor for any adverse effects.
When used appropriately, Tobradex can help the eye heal more comfortably and quickly, but it should never be used longer than advised. Always adhere to the duration recommended by your clinician and the product leaflet.
Before using Tobradex, wash your hands thoroughly. Check the bottle for any signs of damage or contamination. Do not let the dropper touch your eye or any other surface to prevent contamination.
If you wear contact lenses, ask your clinician whether you should remove them before applying Tobradex and when you can safely reinsert them after use. Do not share your eye drops with others and do not reuse an opened bottle beyond its recommended use period.
To apply Tobradex, tilt your head slightly or lie down, and gently pull down the lower eyelid to create a small pocket. Instill one drop into the pocket of the affected eye. After applying, close the eye gently and avoid pressing on the eye too hard. If advised, you may lightly press your finger at the inner corner of the eye to reduce drainage.
If you are using other eye medications, wait a short time between medicines as directed by your clinician. Do not exceed the amount prescribed or the duration advised by your healthcare professional. If you notice any changes in your vision after use, inform your clinician promptly.
Store Tobradex at room temperature away from light and heat. Keep it out of reach of children. Do not use the bottle if it has changed colour, contains particles, or the seal is broken. Use the product exactly as prescribed or as indicated in the leaflet, and contact a healthcare professional if you have questions about timing, duration, or sequence of eye medications.
You should not use Tobradex if you are known to be allergic to tobramycin, dexamethasone, or any other ingredient in the product. An allergic reaction can be serious and requires medical attention.
Tobradex is not suitable for certain eye infections caused by viruses or fungi unless advised by a clinician. Do not use Tobradex if you have a corneal perforation or if your doctor has advised against steroid use in your eye condition.
If you have a history of glaucoma, cataracts, or other eye conditions, tell your clinician before starting Tobradex. Certain medical conditions or medications can affect how Tobradex works, so full medical history is important.
Special considerations apply to children, pregnancy, and breastfeeding. A clinician will determine whether Tobradex is appropriate in these situations and will provide dosing guidance and monitoring instructions. If you have concerns about these populations, discuss them with your healthcare professional.
Tobradex can cause side effects related to both the antibiotic and the steroid components. Short-term use under supervision minimizes risk, but some people may experience eye irritation, blurred vision, or redness after applying the drops.
Prolonged use of steroid-containing eye drops can increase the risk of elevated intraocular pressure, cataract formation, or secondary infections. If you notice a change in vision, persistent eye pain, or a dull ache around the eye, seek medical advice promptly.
If you have a history of eye injuries, infections, or a compromised immune system, discuss Tobradex use with your clinician. If you develop a severe reaction or a new symptom such as swelling of the face or lips, seek urgent medical help.
Tell your clinician about all medicines you are taking, including other eye drops and any vitamins or supplements. Some medicines can interact with Tobradex or affect how well it works. Never share eye drops with others and do not use drops that are past their expiry date or that have been stored improperly.
Common side effects include a temporary burning, stinging, or blurred vision after application. Some people may also notice tearing or redness in the eye, or a mild sensation of grittiness.
Less common effects can include a dry eye sensation, taste changes, or minor irritation around the eye after use. If you notice continued eye irritation, swelling, or increased redness, contact a clinician for guidance.
Serious side effects are rare but require urgent medical attention. Seek immediate help if you experience severe allergic reactions (such as swelling of the face or throat, hives, or severe dizziness), severe eye pain, vision changes, or an eye injury following use.
Long-term or inappropriate use can increase the risk of infections or eye surface problems. If you have any concerns about side effects, discuss them with your pharmacist or clinician. Do not continue to use Tobradex if you suspect a serious reaction, unless advised by a clinician.
Tell your clinician about all medicines you are taking, including non-prescription eye drops and other ocular medications. Some products may interact with Tobradex or affect how well it works.
If you are using more than one eye drop, spaces between applications are usually recommended. Do not mix eye drops directly or apply them in the same eye at the same time in a manner that could cause contamination of the bottle or reduce effectiveness.
There is a potential for increased side effects when combining Tobradex with other corticosteroid-containing products. Discuss all medications with your healthcare professional to determine the safest plan for your situation.
The safety of Tobradex during pregnancy has not been fully established. It should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus, and under the guidance of a healthcare professional.
Breast-feeding while using Tobradex is not well studied. If you are nursing, talk with your clinician about the potential risks and benefits to decide whether to use Tobradex during this period.
If you become pregnant or plan to become pregnant while using Tobradex, inform your clinician promptly. They can provide guidance on alternatives or adjustments to your treatment plan.
Whether Tobradex is available without a prescription depends on local rules. In many places, Tobradex requires a prescription from a healthcare professional. Check with your pharmacist or doctor for the status in your area.
Many people notice improvement within a few days of starting treatment, but the exact timing can vary. If there is no improvement after a few days or symptoms worsen, contact your clinician for advice.
If you miss a dose, use it as soon as you remember unless it is nearly time for your next dose. Do not double up to make up for a missed dose. If you are unsure what to do, ask your clinician or pharmacist for guidance.
Yes, but you should usually space out the use of different eye drops and follow your clinician’s instructions. Tell your healthcare professional about all medicines you are using to avoid potential interactions.
In most cases, you should remove contact lenses before applying Tobradex. Reinsert them only when advised by your clinician. If you are told you may wear lenses again after use, wait as directed before putting them back in.
Tobradex may be used in children if a clinician determines it is appropriate. The dosing and monitoring will be guided by the child’s age and condition. Do not use Tobradex in a child without medical advice.
Temporary blurred vision or changes in vision can occur after applying Tobradex. Do not drive or operate machinery if your vision is blurred until it clears. If blurred vision persists, contact your clinician.
Seek urgent medical help if you experience symptoms such as swelling of the face or lips, severe rash, itching, or trouble breathing. Allergic reactions require prompt evaluation and treatment.
If you have persistent eye pain, redness that worsens, or eye discharge that does not improve, contact your clinician promptly. These can be signs that the infection or inflammation is not adequately controlled.
Store Tobradex at room temperature, away from heat and light. Keep the bottle tightly closed and out of reach of children. Do not use the product after the expiry date printed on the label.
Follow-up with your clinician is often advised to confirm that the infection or inflammation has resolved and to check for any side effects. Ask your clinician how long you should expect to continue monitoring your eye condition.
The information provided here is based on standard patient information for Tobradex and common clinical guidelines for ocular use. It is intended to support conversations with your healthcare professional.
Official product information and labeling supplied by the manufacturer, as well as Health Canada patient information leaflets for Tobradex, are used to inform general guidance. Your clinician may provide additional instructions tailored to your situation.
For further reading, consult your pharmacist or doctor, and refer to the product leaflet that accompanies the medication you receive. Always rely on professional medical advice for diagnosis and treatment choices.
Clinical practice guidelines from Canadian ophthalmology resources may offer background on the management of ocular infections and inflammation. Your healthcare professional can interpret these in the context of your health history and current condition.
14–21 days. Free from C$275.68 .
5–9 days. C$41.35
−10% when paying with cryptocurrency.
−10% on all repeat orders.
All orders are packed in neutral, unbranded boxes with no product name on the outside.
